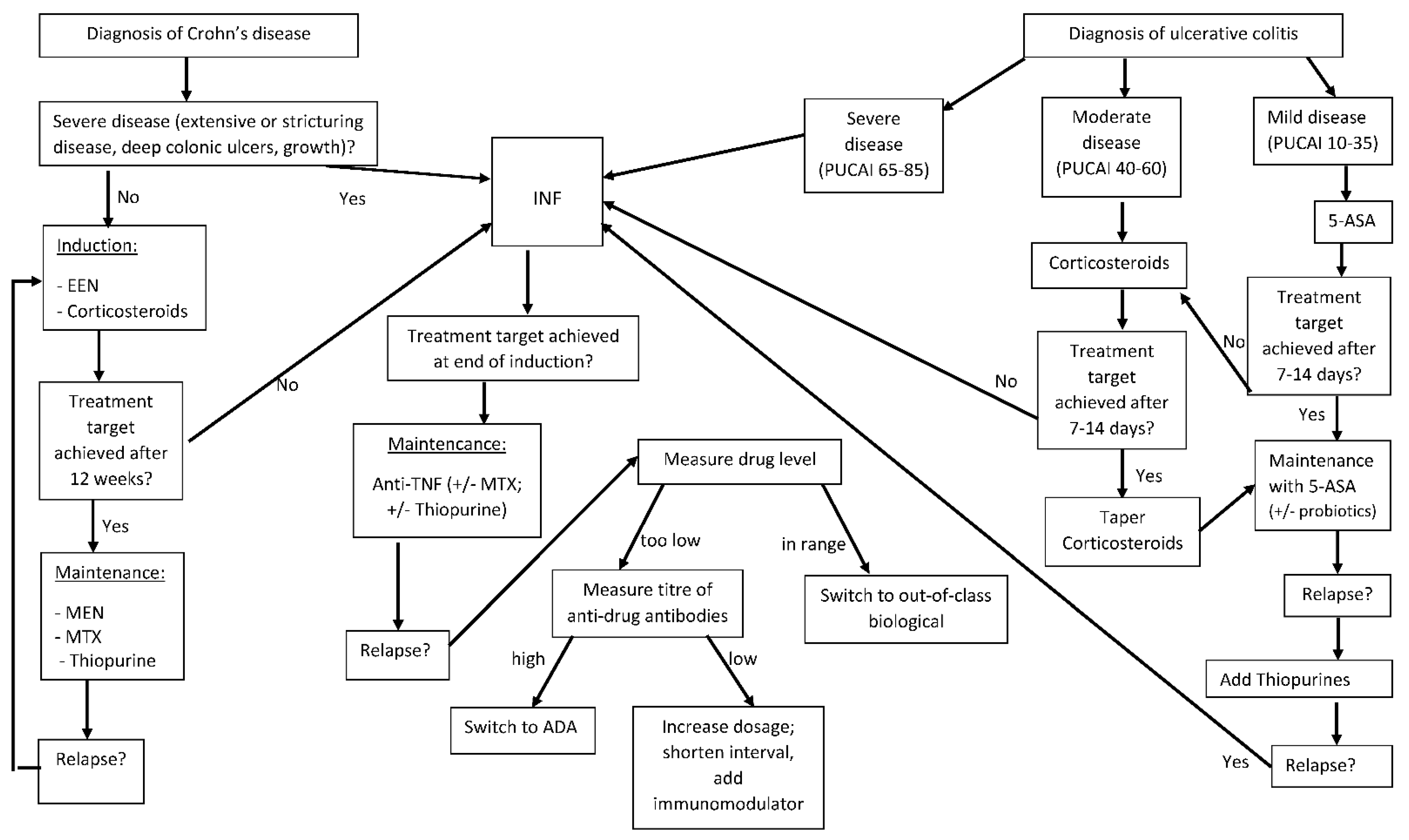
Children | Free Full-Text | Therapy Strategies for Children Suffering from Inflammatory Bowel Disease (IBD)—A Narrative Review | HTML

PDF) Biologics recommendations in the ECCO guidelines on therapeutics in Crohn's disease: Medical treatment
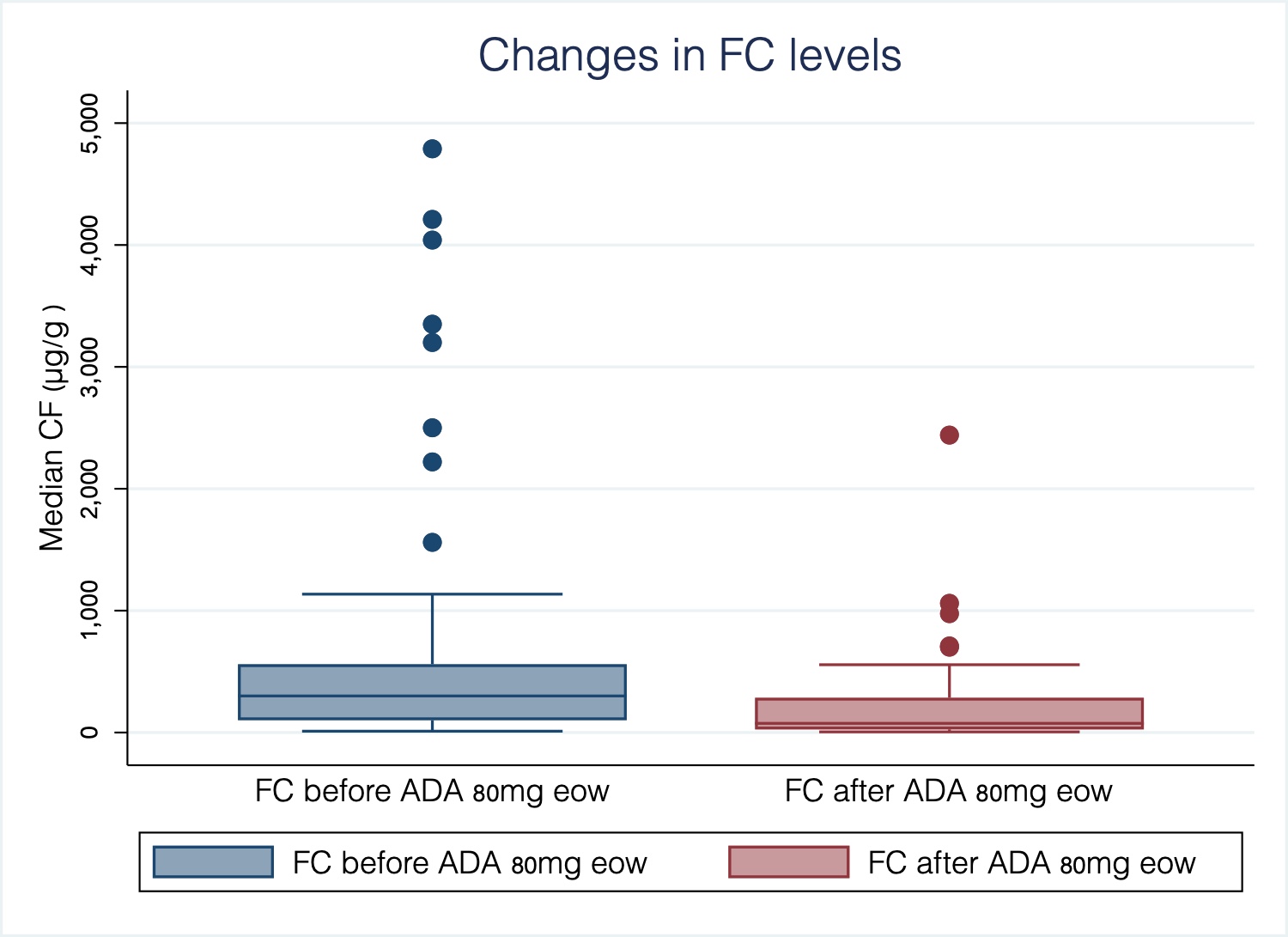
European Crohn´s and Colitis Organisation - ECCO - P533 Adalimumab 80mg every other week in inflammatory bowel disease: Treatment intensification outcomes in real life clinical practice
ECCO Guidelines on the Prevention, Diagnosis, and Management of Infections in Inflammatory Bowel Disease
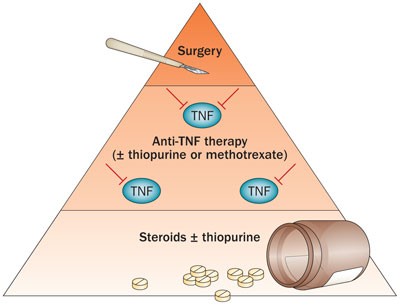
First-line therapy in adult Crohn's disease: who should receive anti-TNF agents? | Nature Reviews Gastroenterology & Hepatology
3rd European Evidence-based Consensus on the Diagnosis and Management of Crohn's Disease 2016: Part 1: Diagnosis and Medical M
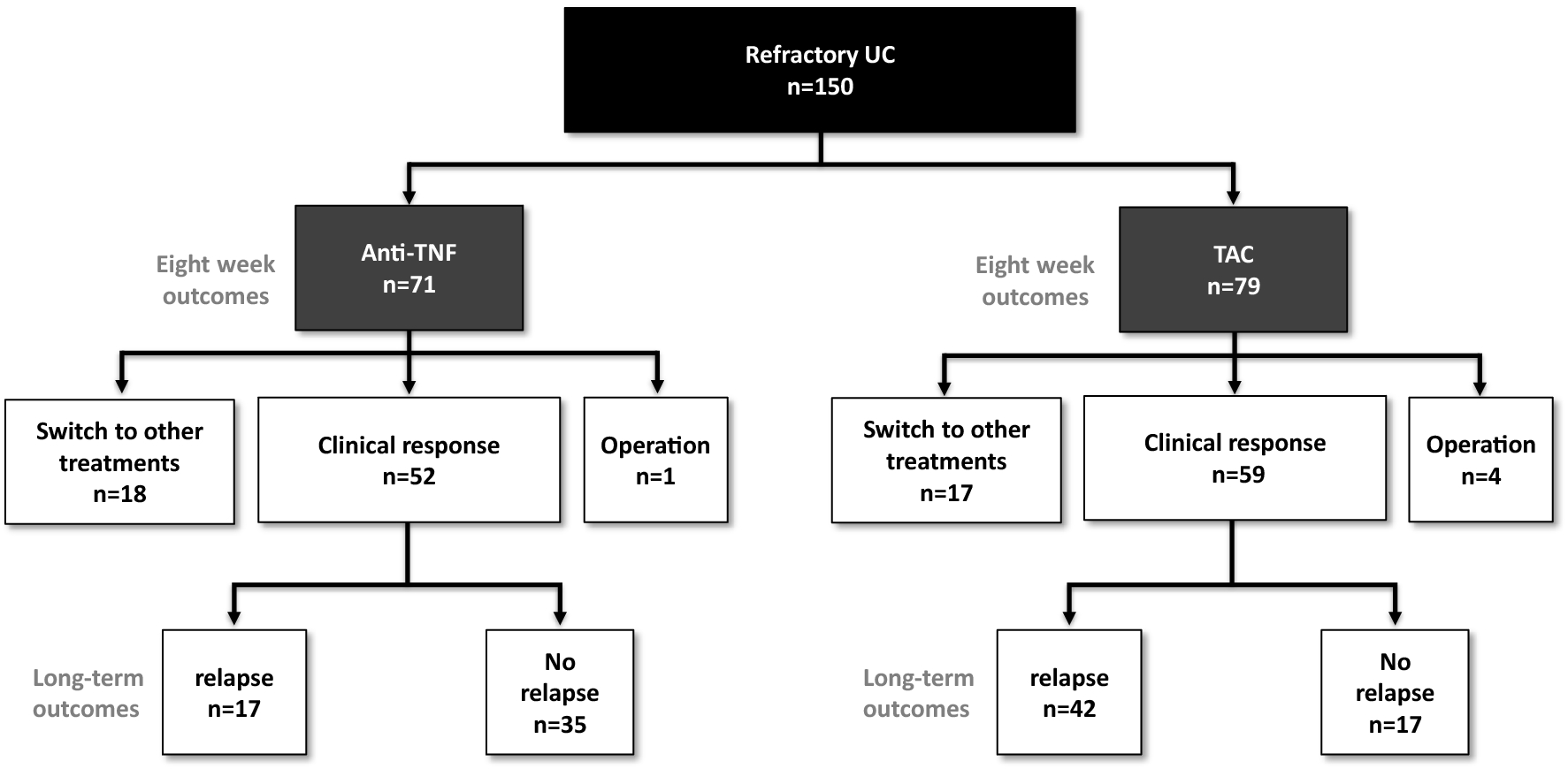
Comparative efficacy of antitumor necrosis factor agents and tacrolimus in naïve steroid-refractory ulcerative colitis patients | Scientific Reports

Proactive Monitoring of Adalimumab Trough Concentration Associated With Increased Clinical Remission in Children With Crohn's Disease Compared With Reactive Monitoring - Gastroenterology

European Crohn´s and Colitis Organisation - ECCO - DOP62 Immunogenicity to second anti-TNF therapy (IMSAT): Implications for sequencing of biologic therapy
Best practices on immunomodulators and biologic agents for ulcerative colitis and Crohn's disease in Asia

Proactive therapeutic drug monitoring of adalimumab for pediatric Crohn's disease patients: A cost‐effectiveness analysis - Yao - 2021 - Journal of Gastroenterology and Hepatology - Wiley Online Library
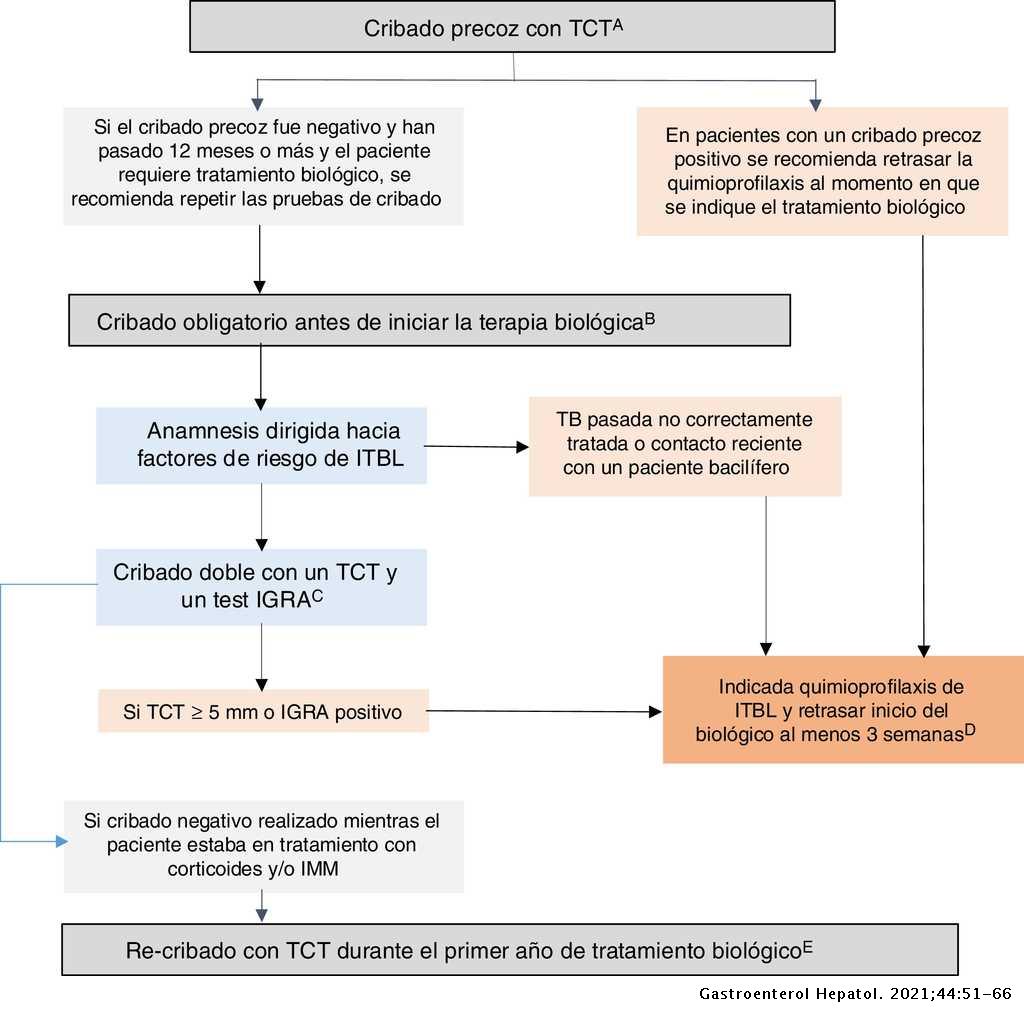
Recommendations of the Spanish Working Group on Crohn's Disease and Ulcerative Colitis (GETECCU) on screening and treatment of tuberculosis infection in patients with inflammatory bowel disease | Gastroenterología y Hepatología (English Edition)
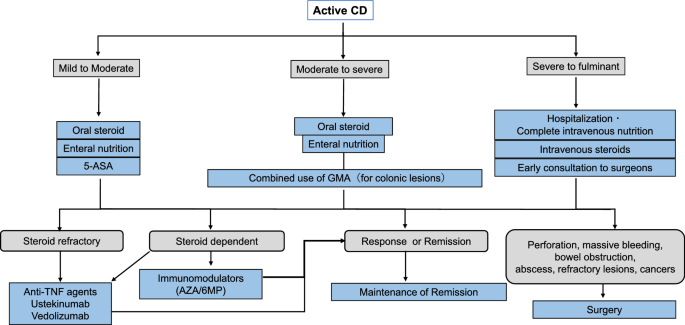
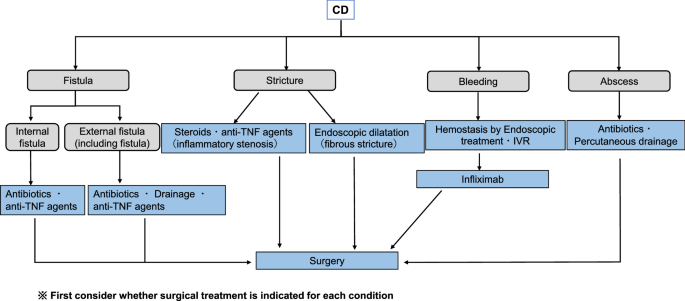
Treatment Algorithms for Crohn's Disease - FullText - Digestion 2020, Vol. 101, Suppl. 1 - Karger Publishers

Efficacy and safety of adalimumab in paediatric patients with moderate-to-severe ulcerative colitis (ENVISION I): a randomised, controlled, phase 3 study - The Lancet Gastroenterology & Hepatology
![PDF] [Comparison of Treatment Guidelines for Ulcerative Colitis: Role of Biologics]. | Semantic Scholar PDF] [Comparison of Treatment Guidelines for Ulcerative Colitis: Role of Biologics]. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0a8e924d287b22861a1cb440f147aa0618dbc982/2-Table1-1.png)
PDF] [Comparison of Treatment Guidelines for Ulcerative Colitis: Role of Biologics]. | Semantic Scholar
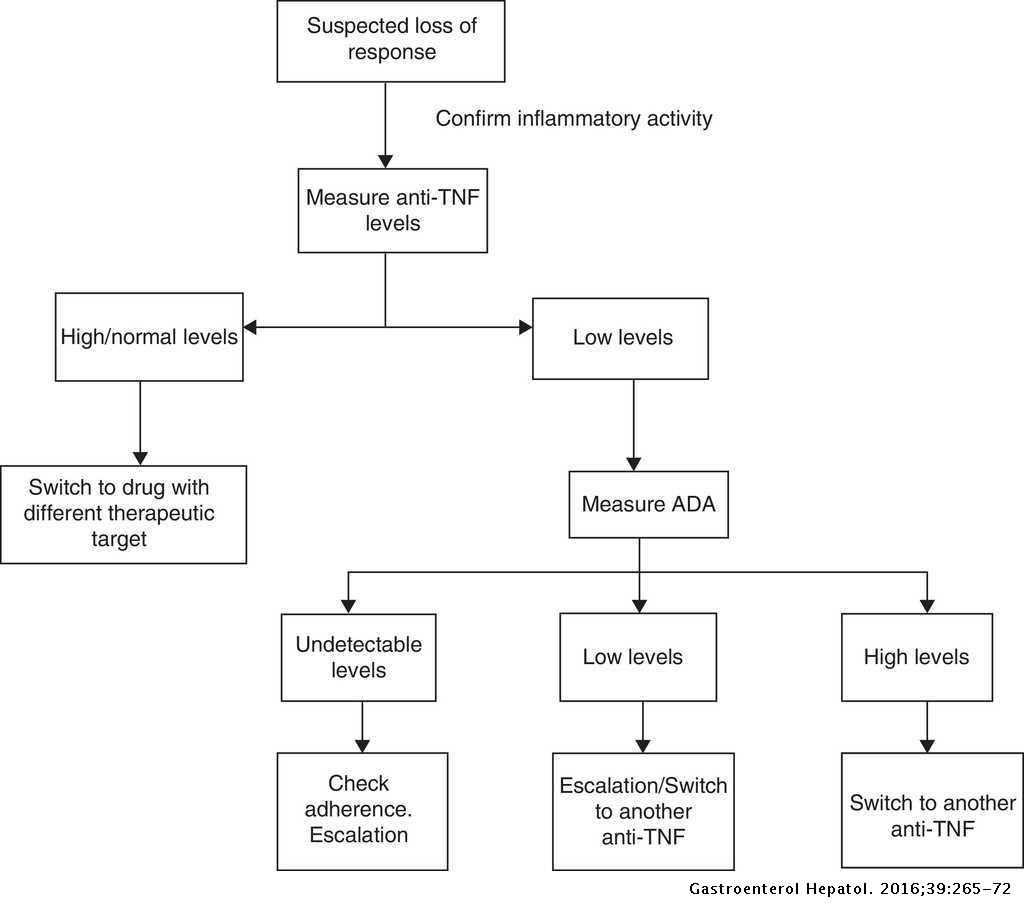
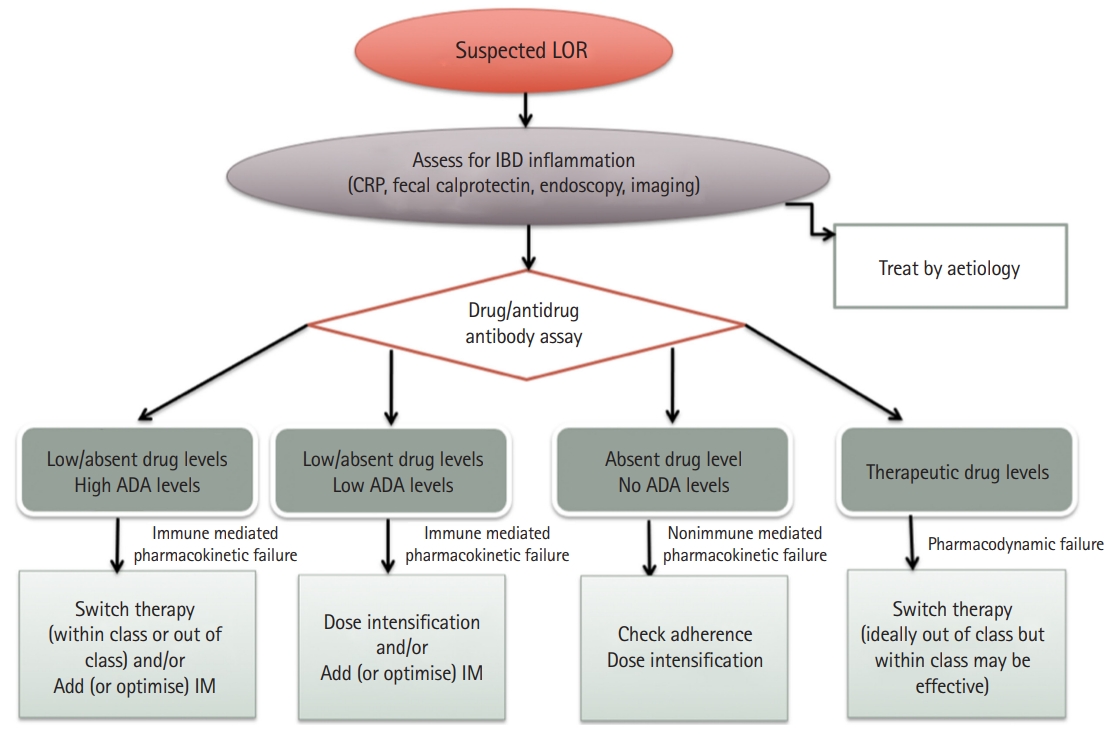
Drugs and anti-drug antibody levels in the management of patients with inflammatory bowel disease | Gastroenterología y Hepatología (English Edition)

PDF) Adalimumab vs Azathioprine in the Prevention of Postoperative Crohn's Disease Recurrence. A GETECCU Randomised Trial
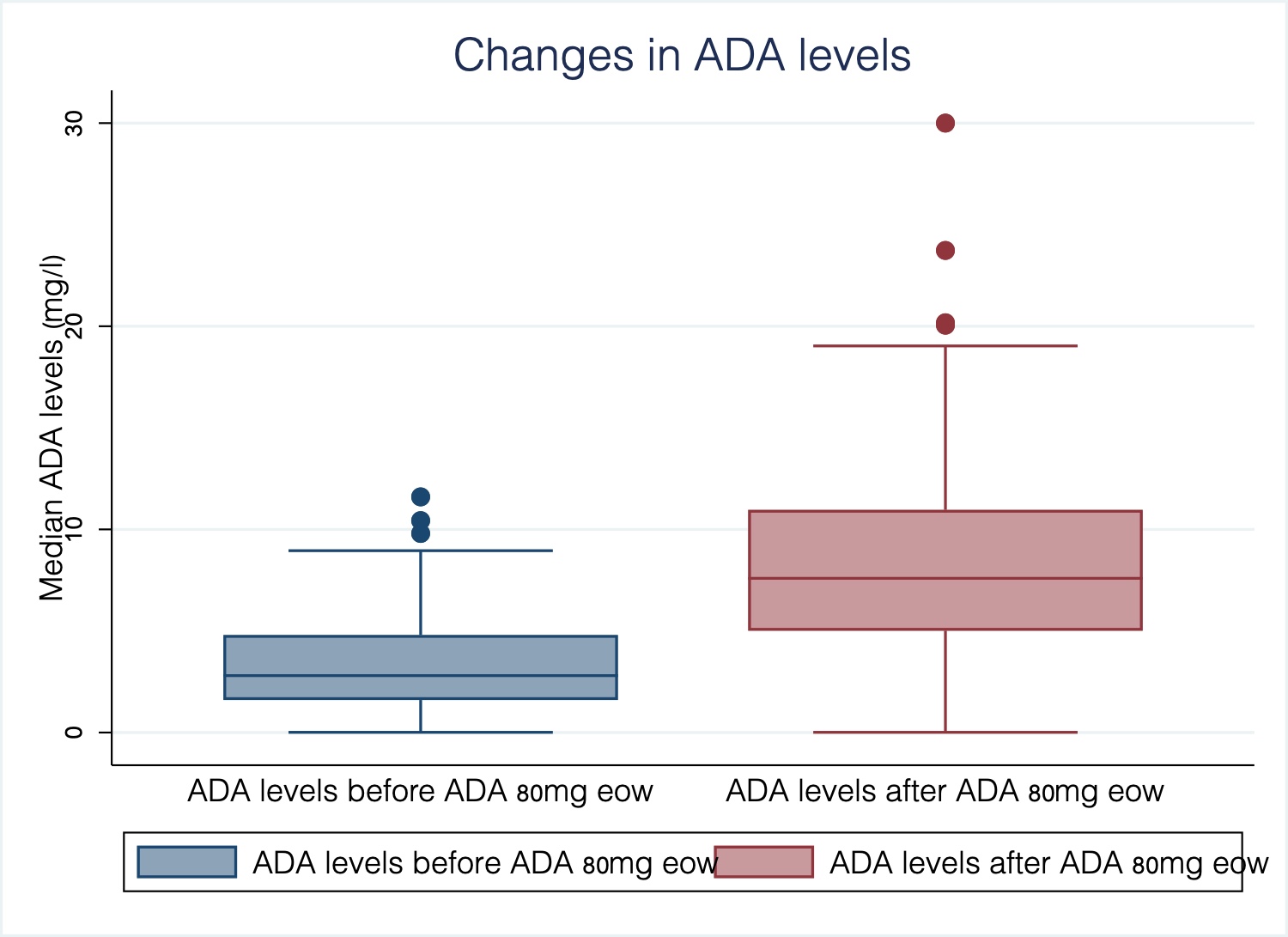
European Crohn´s and Colitis Organisation - ECCO - P533 Adalimumab 80mg every other week in inflammatory bowel disease: Treatment intensification outcomes in real life clinical practice
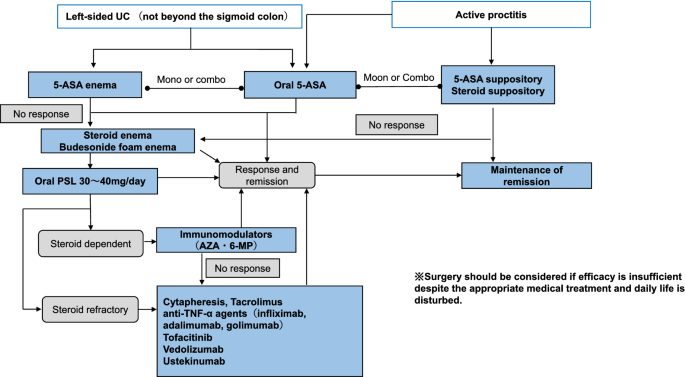
Third European Evidence-based Consensus on Diagnosis and Management of Ulcerative Colitis. Part 1: Definitions, Diagnosis, Extra