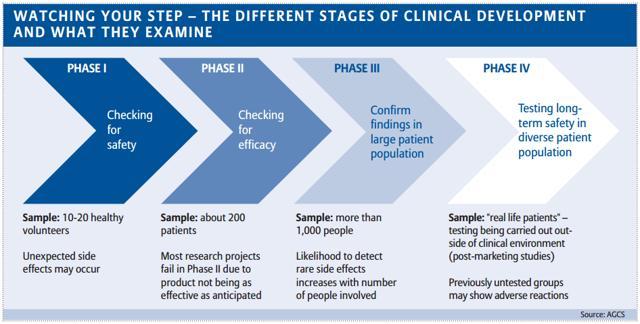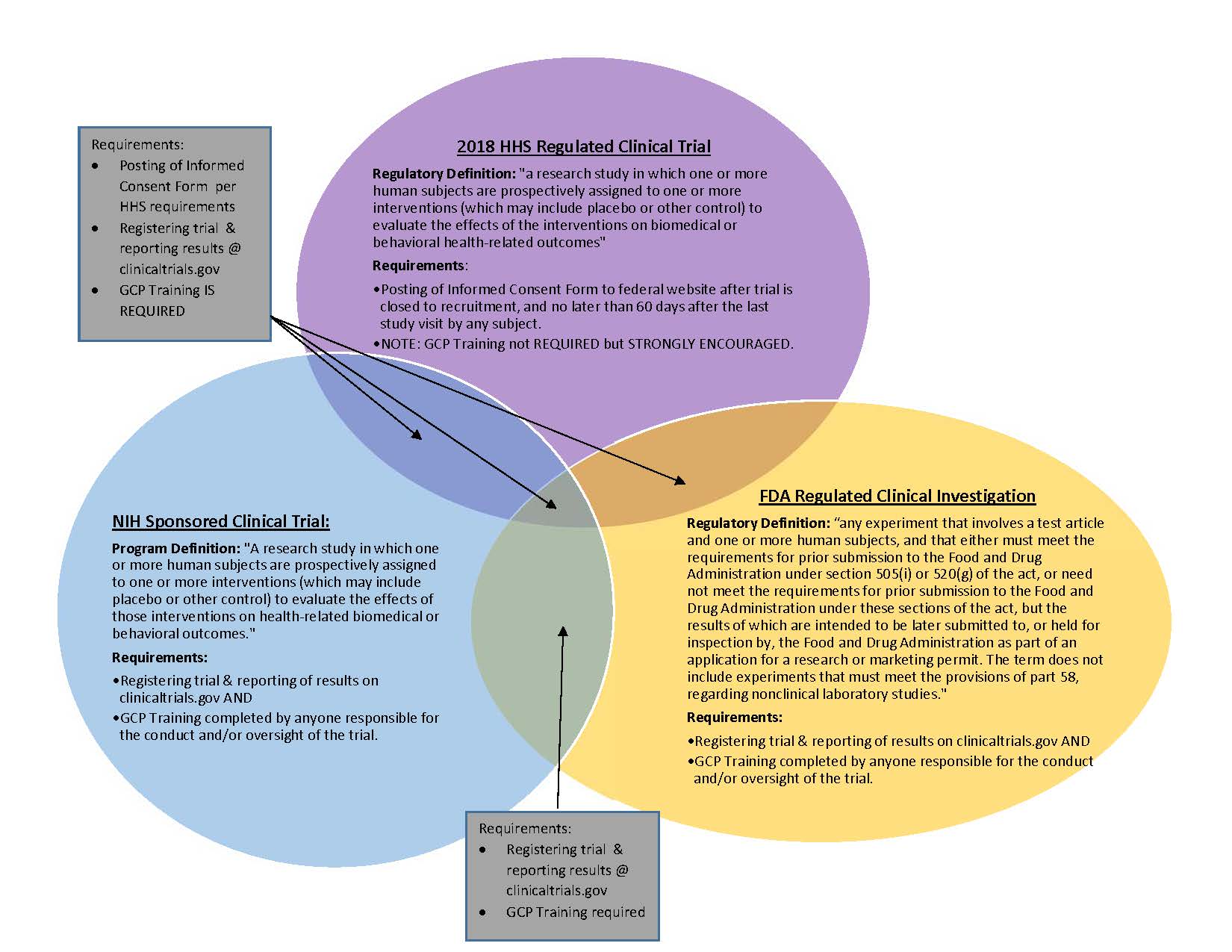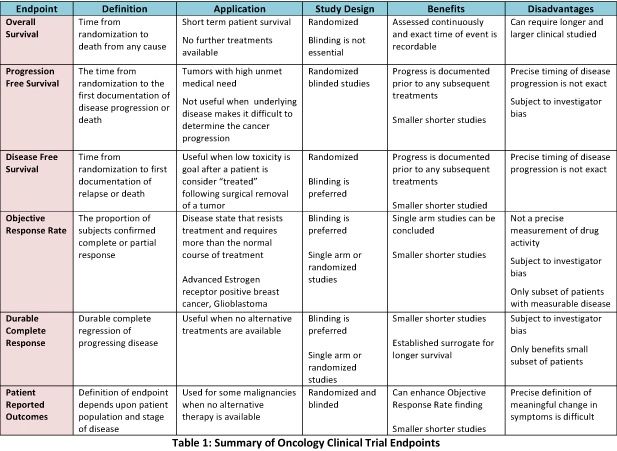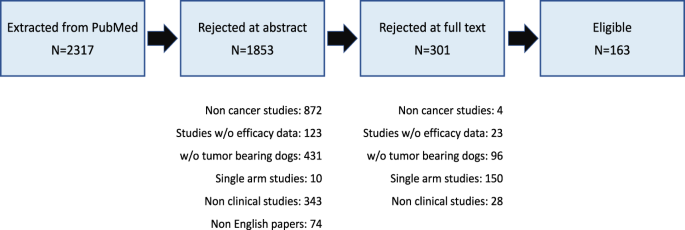
An empirical assessment of research practices across 163 clinical trials of tumor-bearing companion dogs | Scientific Reports

A randomized double-blind placebo-controlled clinical trial of nitazoxanide for treatment of mild or moderate COVID-19 - eClinicalMedicine

Observed and expected serious adverse event rates in randomised clinical trials for hypertension: an observational study comparing trials that do and do not focus on older people - The Lancet Healthy Longevity

Cardiovascular outcomes in adults with hypertension with evening versus morning dosing of usual antihypertensives in the UK (TIME study): a prospective, randomised, open-label, blinded-endpoint clinical trial - The Lancet

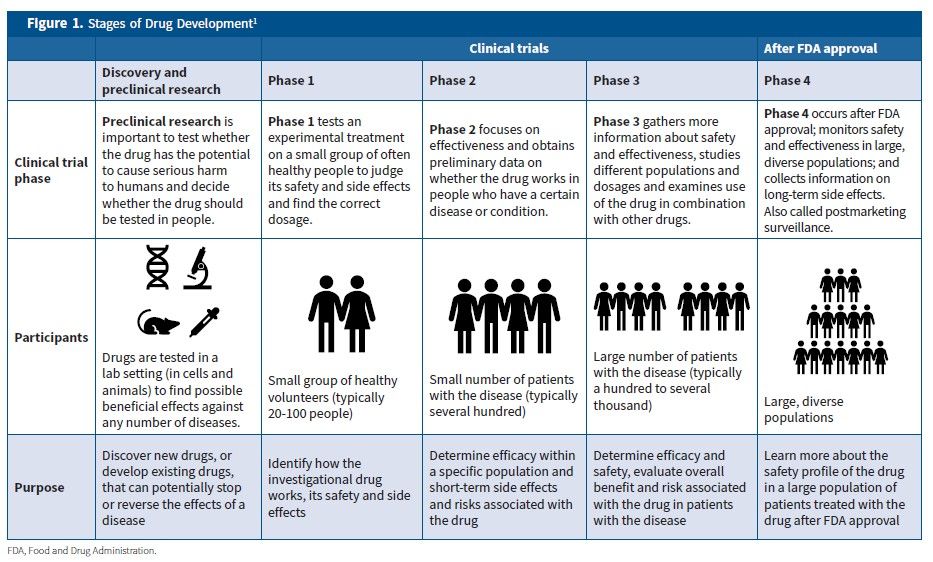
Frontiers | Trends of Phase I Clinical Trials of New Drugs in Mainland China Over the Past 10 Years (2011–2020)

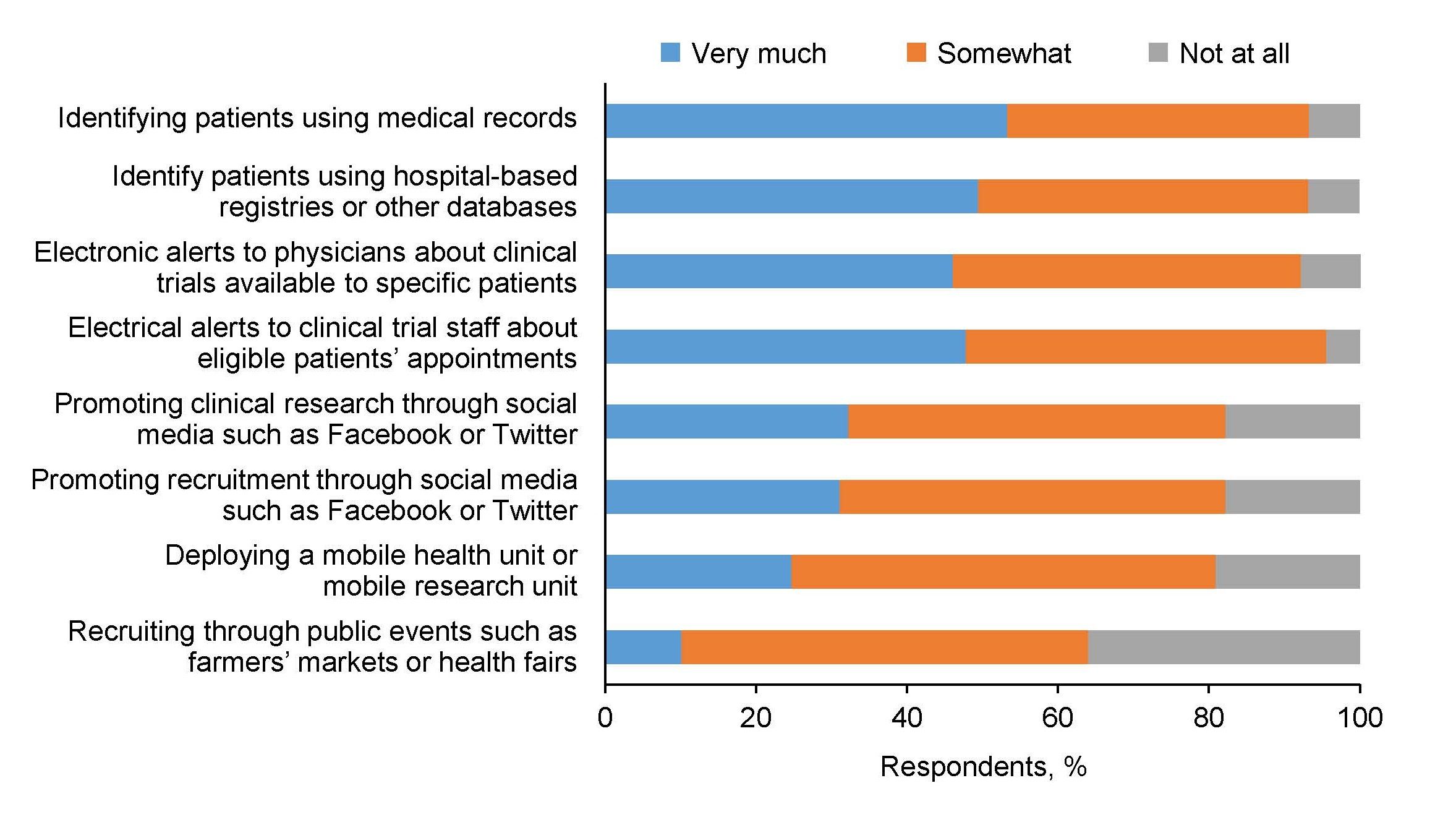
Recruitment of Black Adults into Cardiovascular Disease Trials | Journal of the American Heart Association
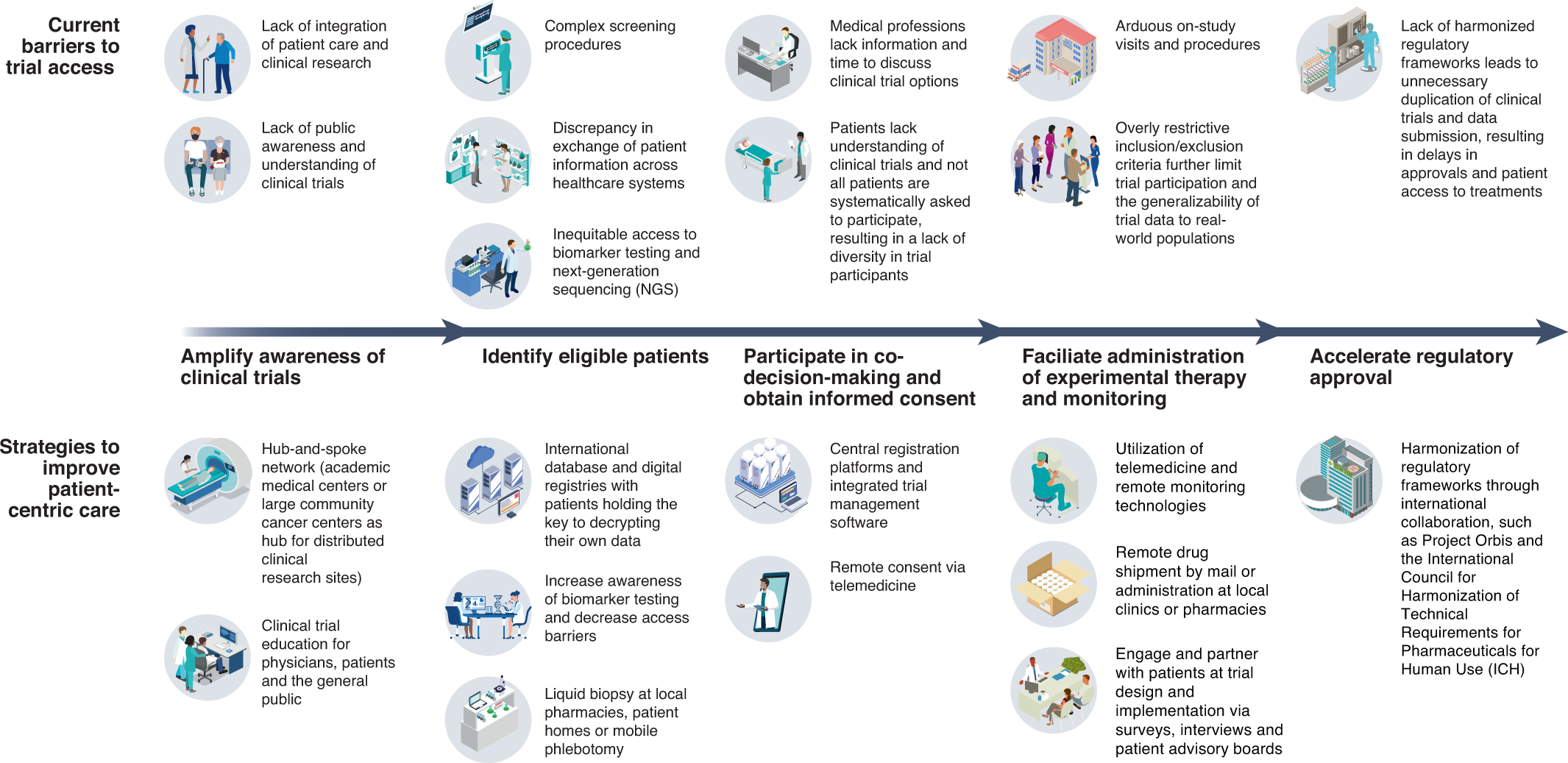
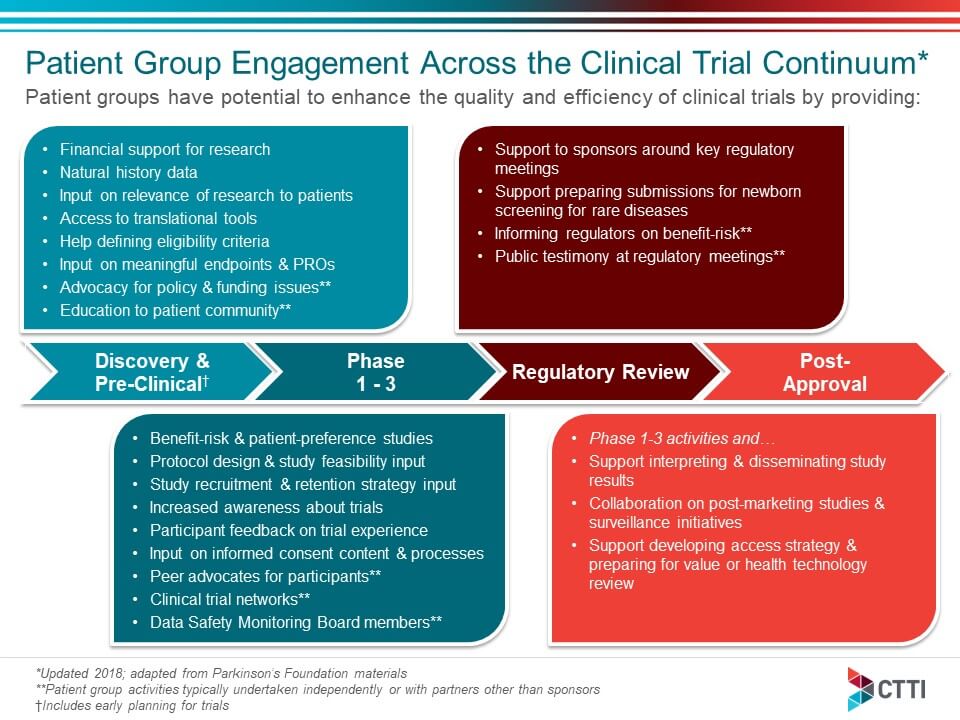
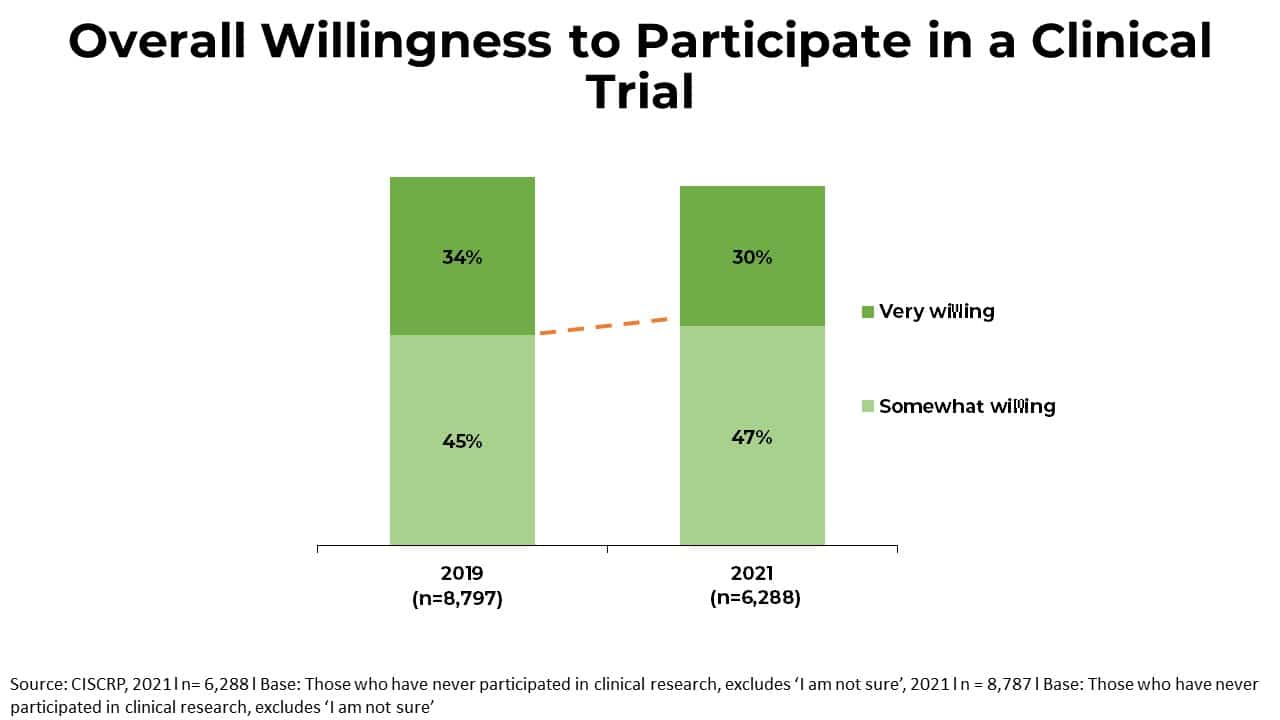
Reimagining patient-centric cancer clinical trials: a multi-stakeholder international coalition | Nature Medicine
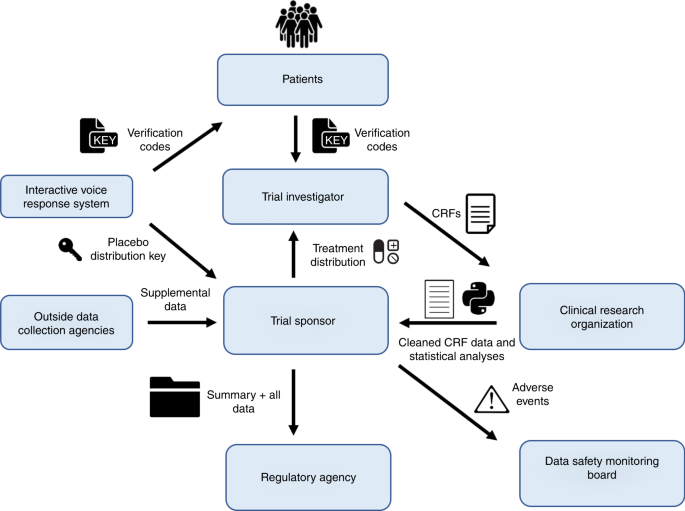
Prototype of running clinical trials in an untrustworthy environment using blockchain | Nature Communications