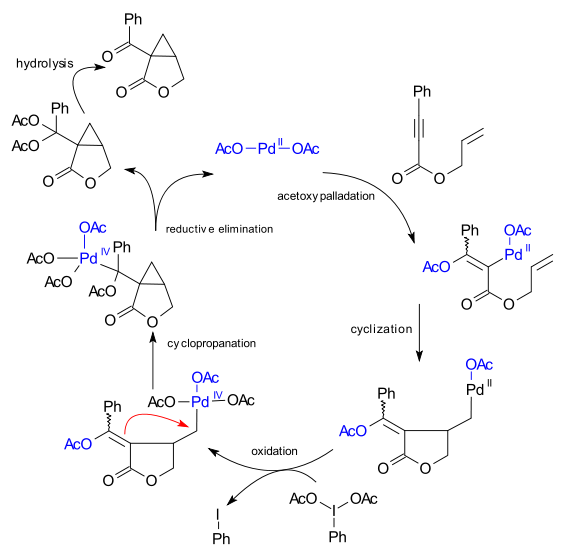
DFT studies of isomerization in palladium(IV) chemistry and alkyl halide transfer from palladium(IV) to palladium(II) - ScienceDirect
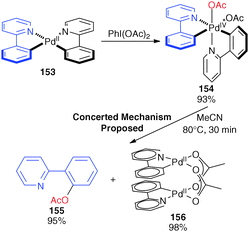
Experiment and computation: a combined approach to study the reactivity of palladium complexes in oxidation states 0 to iv - Chemical Society Reviews (RSC Publishing)

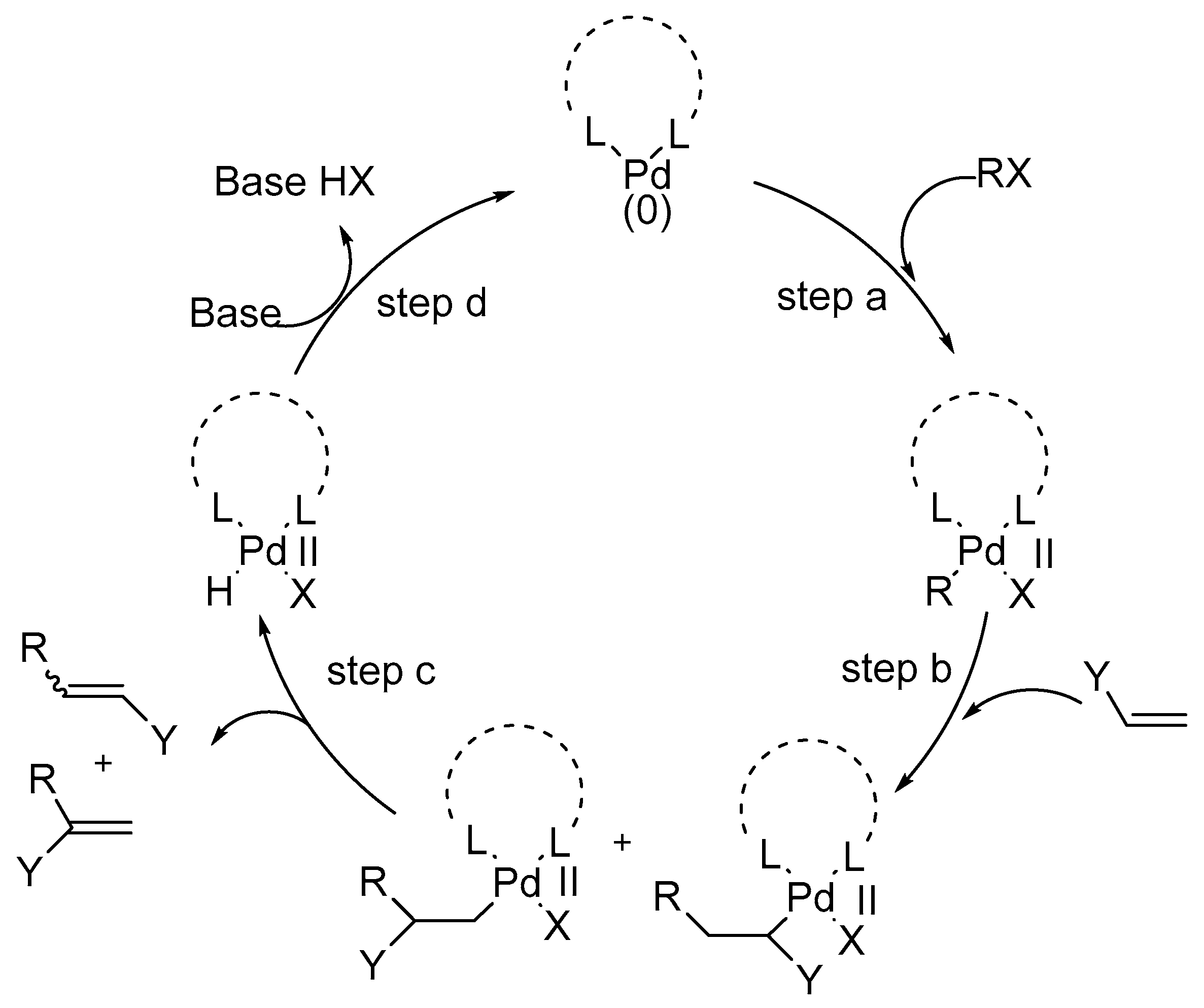
Report: Toward Greater Understanding and Expanded Utility of the Palladium-Catalyzed Activation of Carbon-Carbon Single Bonds (56th Annual Report on Research Under Sponsorship of The American Chemical Society Petroleum Research Fund)
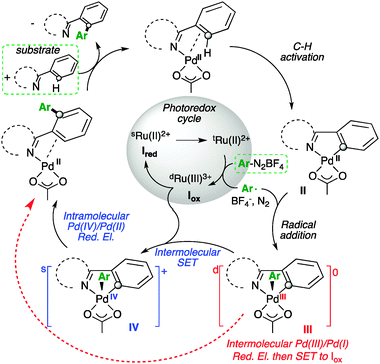
Radical Pd(iii)/Pd(i) reductive elimination in palladium sequences - Chemical Communications (RSC Publishing)

Proposed mechanism for Pd-catalysed oxidative coupling of 2aryl pyridines. | Download Scientific Diagram

Scheme 1: Mechanism of palladium (II)-catalyzed oxidations of leucine... | Download Scientific Diagram

Mechanism of the palladium(II/IV)-catalysed intermolecular C–H amination. | Download Scientific Diagram
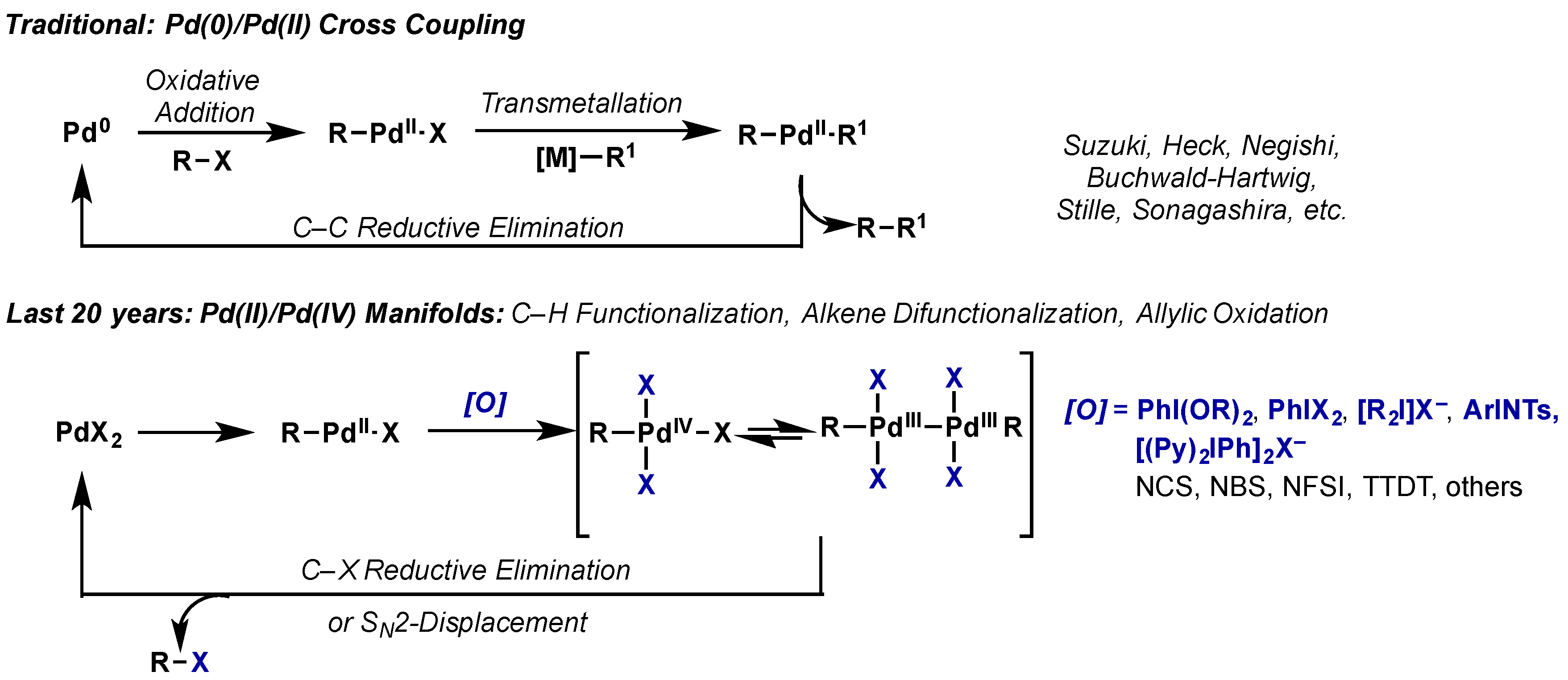
Molecules | Free Full-Text | Hypervalent Iodine Reagents in High Valent Transition Metal Chemistry | HTML

Figure 10 from Mechanism of C-F reductive elimination from palladium(IV) fluorides. | Semantic Scholar

Figure 7 from The mechanism of a ligand-promoted C(sp3)-H activation and arylation reaction via palladium catalysis: theoretical demonstration of a Pd(II)/Pd(IV) redox manifold. | Semantic Scholar

Ligand‐Assisted Palladium(II)/(IV) Oxidation for sp3 CH Fluorination - Sun - 2016 - Advanced Synthesis & Catalysis - Wiley Online Library

Figure 1 from The mechanism of a ligand-promoted C(sp3)-H activation and arylation reaction via palladium catalysis: theoretical demonstration of a Pd(II)/Pd(IV) redox manifold. | Semantic Scholar

Figure 3 from The mechanism of a ligand-promoted C(sp3)-H activation and arylation reaction via palladium catalysis: theoretical demonstration of a Pd(II)/Pd(IV) redox manifold. | Semantic Scholar