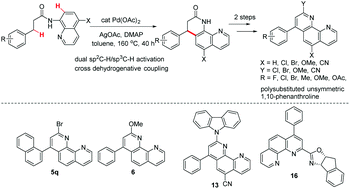
Practical synthesis of polysubstituted unsymmetric 1,10-phenanthrolines by palladium catalyzed intramolecular oxidative cross coupling of C(sp2)–H and C(sp3)–H bonds of carboxamides - Organic Chemistry Frontiers (RSC Publishing)
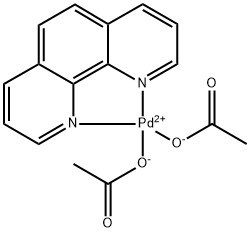
Acetato(2,9-dimethyl-1,10-phenanthroline)palladium(II) dimer bis(trifluoromethanesulfonate), 99% CAS#: 959698-19-2

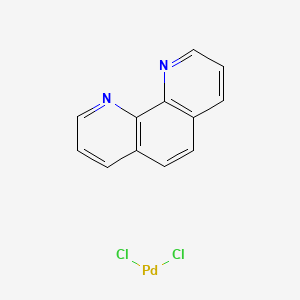
China CAS 14783-10-9 Dichloro (1, 10-phenanthroline) Palladium (II) C12h8cl2n2pd - China Palladium Catalyst, Ruthenium Catalyst

Crystal structure of diiodo(1,10-phenanthroline-κ2N,N′)palladium(II), PdI2(C12H8N2) : Zeitschrift für Kristallographie - New Crystal Structures

Palladium‐Catalyzed Intramolecular Cyclization of Nitroalkenes: Synthesis of Thienopyrroles - El‐Atawy - 2017 - European Journal of Organic Chemistry - Wiley Online Library

Design and synthesis of chiral 1,10-phenanthroline ligand, and application in palladium catalyzed asymmetric 1,4-addition reactions - ScienceDirect

PDF) Friedel–Crafts-Type Alkylation of Indoles in Water Using Amphiphilic Resin-Supported 1,10-Phenanthroline–Palladium Complex under Aerobic Conditions
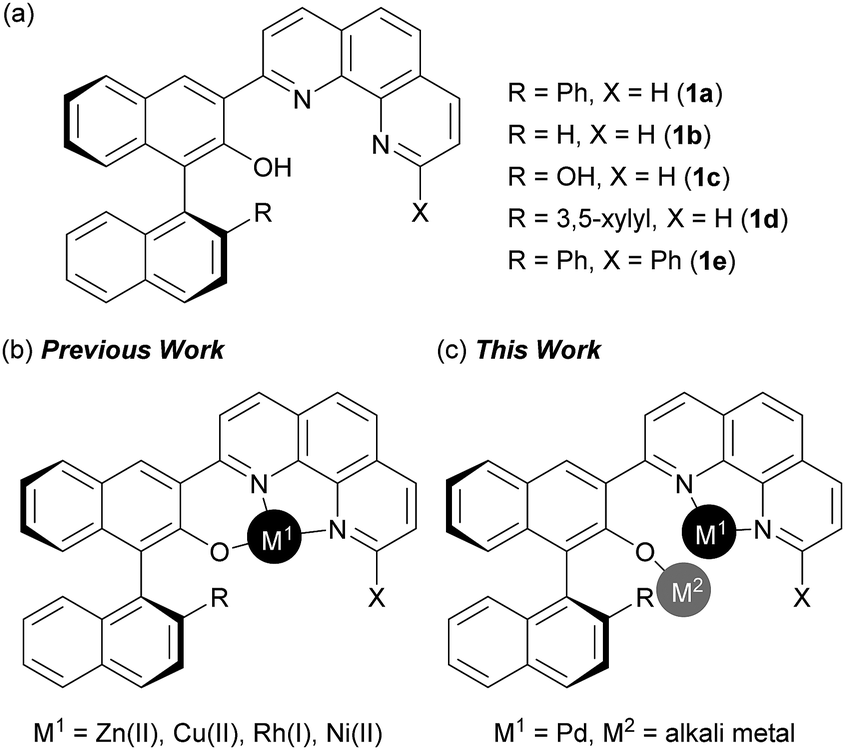
Design of bifunctional chiral phenanthroline ligand with Lewis basic site for palladium-catalyzed asymmetric allylic substitution - Chemical Communications (RSC Publishing) DOI:10.1039/C8CC00754C
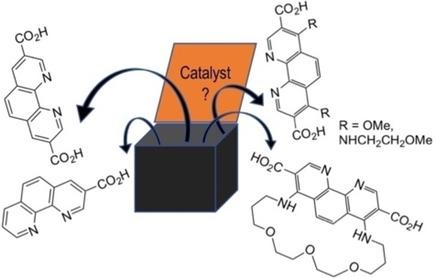
1,10‐Phenanthroline Carboxylic Acids for Preparation of Functionalized Metal‐Organic Frameworks - Asian J. Org. Chem. - X-MOL
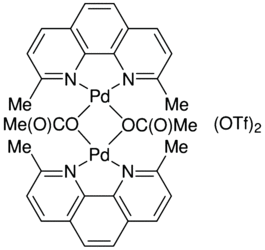
1,10-Phenanthroline, 2,9-dimethyl-4,7-diphenyl-, palladium salt, acetate (1:1:2) | C30H28N2O4Pd | ChemSpider