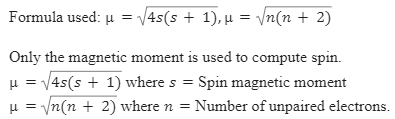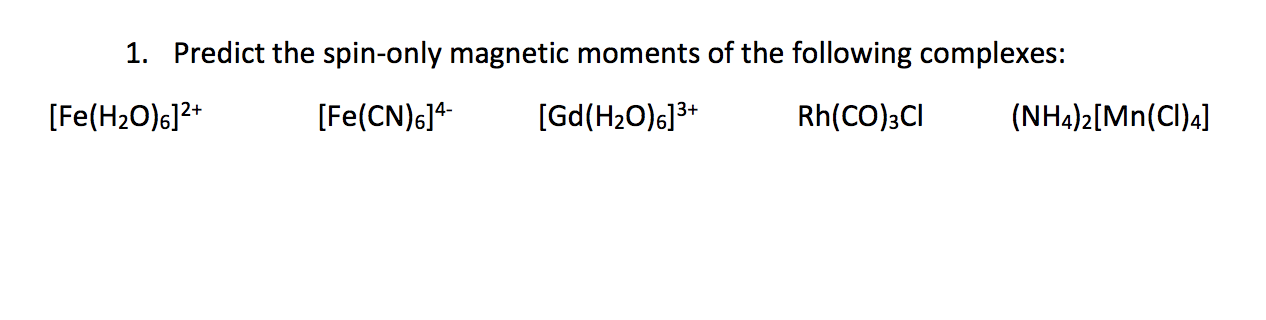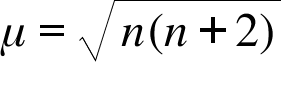Spin only magnetic moment of `._(25)Mn^(x+)`ion is `sqrt(15)` B.M. Then, the value of `x` is `:` - YouTube
What is the spin-only magnetic moment value (BM) of a divalent metal ion with atomic number 25, in - Sarthaks eConnect | Largest Online Education Community

Solve this: Q 85 The spin only magnetic moment value ( in B M units) of CrCO6 is - Chemistry - The d-and f-Block Elements - 12561515 | Meritnation.com
Calculate the spin only magnetic moment of La^3+. - Sarthaks eConnect | Largest Online Education Community
![The spin only magnetic moment value of `[MnBr_(4)]^(2-)` ion is `5.9BM` On the basis of `VBT` - YouTube The spin only magnetic moment value of `[MnBr_(4)]^(2-)` ion is `5.9BM` On the basis of `VBT` - YouTube](https://i.ytimg.com/vi/no_ZGBD689M/maxresdefault.jpg)
The spin only magnetic moment value of `[MnBr_(4)]^(2-)` ion is `5.9BM` On the basis of `VBT` - YouTube
The value of the 'spin only' magnetic moment for one of the following configurations is 2.84 BM. The correct one is - Sarthaks eConnect | Largest Online Education Community