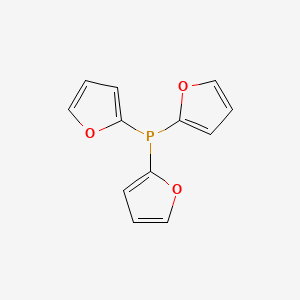
![Tris[tri(2‐thienyl)phosphine]palladium as the catalyst precursor for thiophene‐based Suzuki‐Miyaura crosscoupling and polycondensation - Li - 2008 - Journal of Polymer Science Part A: Polymer Chemistry - Wiley Online Library Tris[tri(2‐thienyl)phosphine]palladium as the catalyst precursor for thiophene‐based Suzuki‐Miyaura crosscoupling and polycondensation - Li - 2008 - Journal of Polymer Science Part A: Polymer Chemistry - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/4dbbd39f-04c1-40c9-be16-0a73bbc6236d/mgra001.jpg)
Tris[tri(2‐thienyl)phosphine]palladium as the catalyst precursor for thiophene‐based Suzuki‐Miyaura crosscoupling and polycondensation - Li - 2008 - Journal of Polymer Science Part A: Polymer Chemistry - Wiley Online Library

CHAPTER 1 New Chemistry for Organic Photovoltaic Materials (RSC Publishing) DOI:10.1039/9781782622307-00001
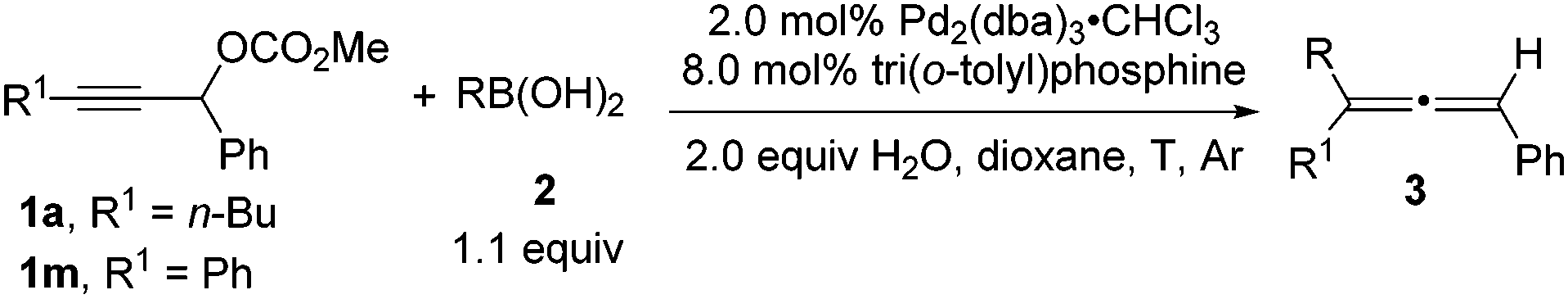
Tri(o-tolyl)phosphine for highly efficient Suzuki coupling of propargylic carbonates with boronic acids - Chemical Communications (RSC Publishing)

Resorcinarene‐Based o‐Biarylphosphines in Palladium‐Catalysed Suzuki–Miyaura Cross‐Coupling Reactions of Bulky Substrates - Elaieb - 2017 - European Journal of Inorganic Chemistry - Wiley Online Library

Resorcinarene‐Based o‐Biarylphosphines in Palladium‐Catalysed Suzuki–Miyaura Cross‐Coupling Reactions of Bulky Substrates - Elaieb - 2017 - European Journal of Inorganic Chemistry - Wiley Online Library

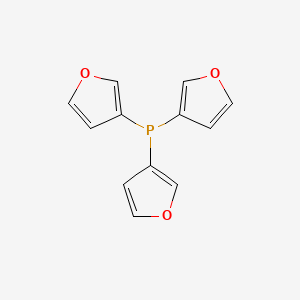
Carbonylation of terminal alkynes catalysed by Pd complexes in combination with tri(2-furyl)phosphine and methanesulfonic acid - ScienceDirect
![5518-52-5・Tri(2-furyl)phosphine・202-18631・208-18633[Detail Information] | [Synthesis & Materials] |Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation 5518-52-5・Tri(2-furyl)phosphine・202-18631・208-18633[Detail Information] | [Synthesis & Materials] |Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation](https://labchem-wako.fujifilm.com/sc/01/5518-52-5.png)
5518-52-5・Tri(2-furyl)phosphine・202-18631・208-18633[Detail Information] | [Synthesis & Materials] |Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation
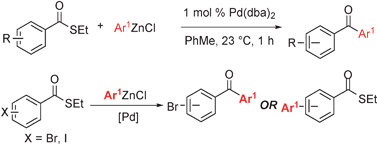
Synthesis of diaryl ketonesvia a phosphine-free Fukuyama reaction - Chemical Communications (RSC Publishing)
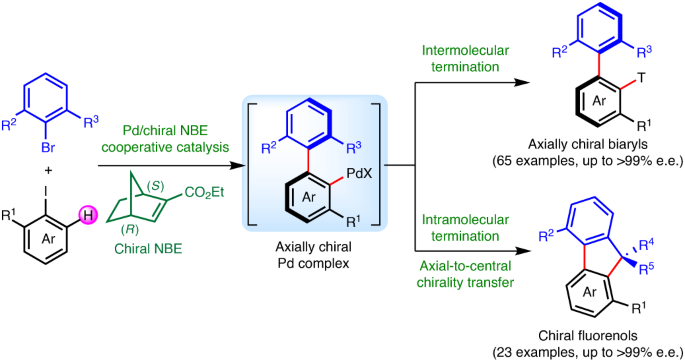
Construction of axial chirality via palladium/chiral norbornene cooperative catalysis | Nature Catalysis
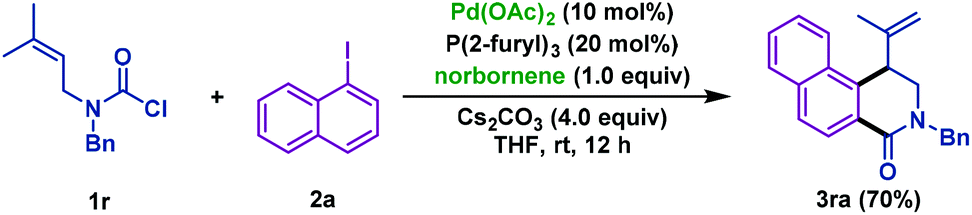
Pd/NBE-catalyzed sequential carbamoylation/olefination of aryl iodides - Organic Chemistry Frontiers (RSC Publishing)