PDF) Direct C-3-Alkenylation of Quinolones via Palladium-Catalyzed CH Functionalization | Mingzong Li - Academia.edu
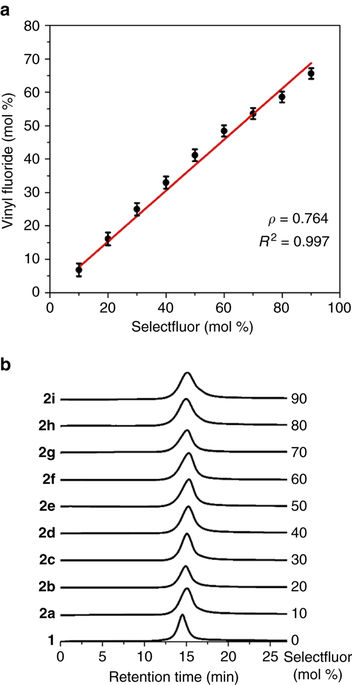
Controlled radical fluorination of poly(meth)acrylic acids in aqueous solution. - Nat. Commun. - X-MOL

PDF) Reaction-Activated Palladium Catalyst for Dehydrogenation of Substituted Cyclohexanones to Phenols and H2 without Oxidants and Hydrogen Acceptors
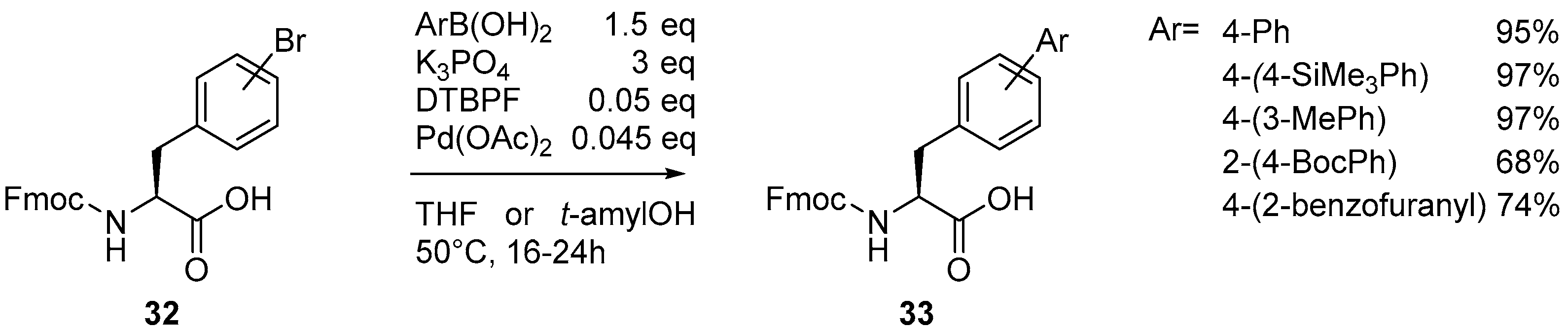
Catalysts | Free Full-Text | The Suzuki–Miyaura Cross-Coupling as a Versatile Tool for Peptide Diversification and Cyclization | HTML

PDF) ChemInform Abstract: Generation of Diverse 2-Pyrones via Palladium- Catalyzed Site-Selective Suzuki-Miyaura Couplings of 3-Bromo-4-tosyloxy-2-pyrone.

A Second‐Coordination‐Sphere Strategy to Modulate Nickel‐ and Palladium‐ Catalyzed Olefin Polymerization and Copolymerization - Li - 2017 - Angewandte Chemie International Edition - Wiley Online Library

Stereoselective Palladium‐Catalyzed Alkenylation and Alkynylation of Thioglycosides - Brachet - 2013 - Advanced Synthesis & Catalysis - Wiley Online Library

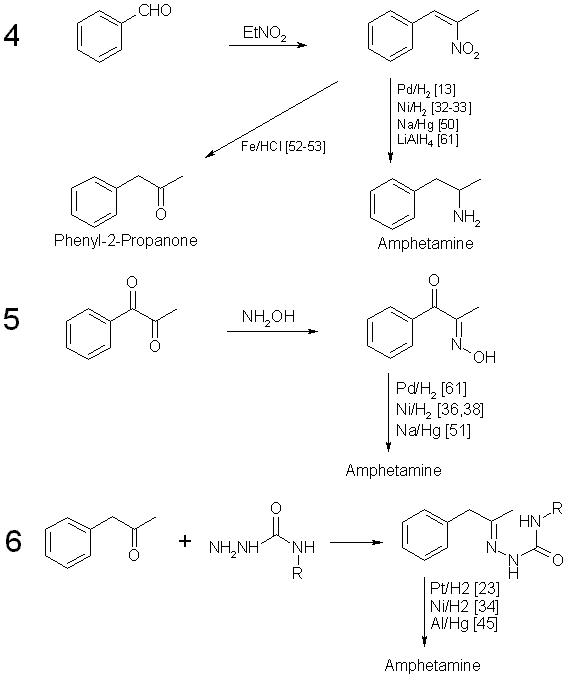
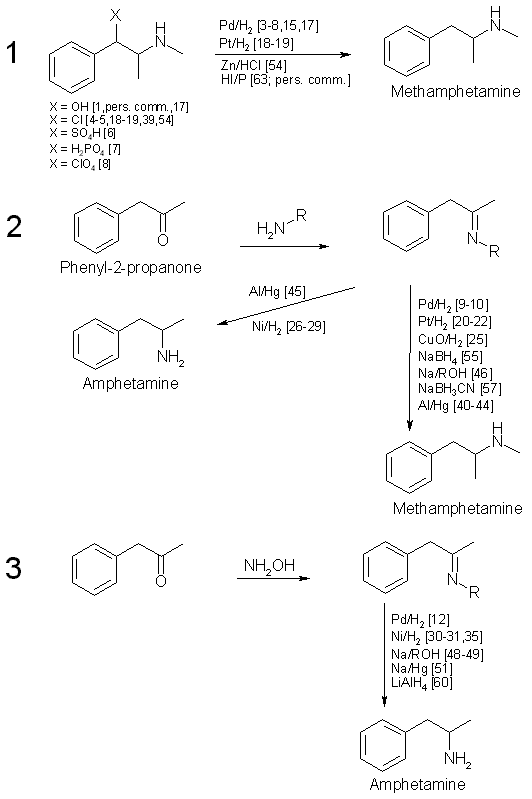
DE60205769T2 - Process for the preparation of amphetamines from phenylpropanolamines - Google Patents
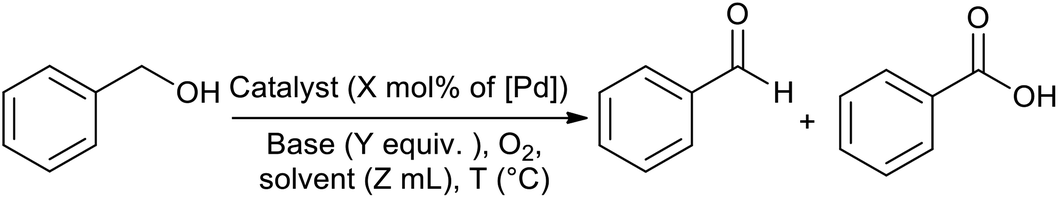
Palladium supported on a novel ordered mesoporous polypyrrole/carbon nanocomposite as a powerful heterogeneous catalyst for the aerobic oxidation of a ... - RSC Advances (RSC Publishing) DOI:10.1039/C9RA10941B

PDF) Synthesis of Functionalized Indoles via Palladium-Catalyzed Cyclization of N-(2-allylphenyl) Benzamide: A Method for Synthesis of Indomethacin Precursor

PDF) Synthesis of Functionalized Indoles via Palladium-Catalyzed Cyclization of N-(2-allylphenyl) Benzamide: A Method for Synthesis of Indomethacin Precursor

PDF) Synthesis of polyamides using palladium-on-carbon (Pd/C) as a heterogeneous, reusable and ligand-free catalytic system

A review of the newly identified impurity profiles in methamphetamine seizures. - Abstract - Europe PMC

Palladium-Catalyzed Alkoxycarbonylation of ( Z )-2-En-4-yn Carbonates Leading to 2,3,5-Trienoates – topic of research paper in Chemical sciences. Download scholarly article PDF and read for free on CyberLeninka open science hub.